

Canadian
maple
Medical Alliance for the Preservation of the Lower Extremity



History of Diabetes
4,700 years ago, (approximately 2670 BCE), in Memphis, (right), the capitol of ancient Egypt, in the reign of the pharaoh Djoser, the first documented record of a symptom of diabetes was recorded.
It was an a papyrus that Hesy-Ra, the world's first
documented physician and dentist, described polyuria,
or frequent urination, accompanying the condition we
know today as diabetes. (1)
Below right is a photograph of a cedar-wood panel
depicting Hesy-Ra, one of several remarkably well-preserved panels found in his tomb in Saqqara, the necropolis of Memphis, just a couple hundred meters
from Djoser's famous stepped pyramid, pictured below. (2)
Djoser's stepped pyramid is the oldest of the Egyptian
pyramids, and as Hesy-Ra lived in this era, we can say
that the first medical description of diabetes can be dated to the first pyramid in Egypt.
Still in Egypt, but over a thousand years later, we
find the Ebers papyrus (right). At 110 pages, dated approximately 1550 BCE, it's the largest compendium of Egyptian medicine known. It describes a large number of medical afflictions and suggested treatments, including intestinal diseases, dentistry, ophthalmology, obstetrics and gynecology, dermatology and the surgical management of abscesses, tumours, fractures and burns.
Diabetes is described as "too great emptying of the urine," for which Egyptians prescribed wheat, fruit, and sweet beer. (3)
(This was probably not great advice, as each of
these suggested treatments would actually tend to elevate sugar levels.)


Let's now move 500 years into the future and over to India.
The origin of Ayurveda (traditional Indian medicine) stretches back as far as 1,000 BCE. In its texts, Indian physicians from this era noted that the urine of diabetics attracted ants and flies. They called the condition "honey urine." As we shall see, this is a name that would be echoed by the west several millennia hence.
The text also noted that diabetic patients suffered from excessive thirst (polydipsia) and bad breath (from ketosis). And it differentiated Type 1 diabetes from Type 2, which it correctly indicated primarily affects the obese and sedentary.
In terms of treatment, there's an emphasis on the role of physical activity to counteract diabetes. (4)
Interestingly, in ancient Greece, there is no record of Hippocrates (460-370 BCE), the Father of Western Medicine, having made mention of the disease.
But back in Egypt, then under the control of Greece following Alexander's conquest in 332 BCE, the physician Apollonius of Memphis, (3rd century BCE, seen to the right) named the condition "diabetes," meaning to pass (-betes) through (dia-). (5)


In Classic Greek, the word 'diabainein' meant to stand with legs apart, as done with urinating.
Interestingly, the word 'diabetes' referred to a drafting compass, left, an instrument that originated in Greece, that had a similar appearance to the legs of a man standing. (6)
It was believed at this time that diabetes was a disease of the kidneys, and physicians at this time recommended bloodletting and dehydration. (5)
In the 1st century CE, it was Aretaeus of Cappadocia, right, a Greek-speaking physician in the then-Roman controlled Egyptian city of Alexandria, who provided the first clinical description of the disease, differentiating between a primary and secondary diabetes.
Aretaeus described the muscle wasting often seen with diabetes rather colorfully as "the melting of flesh and limbs into urine." And he described a diabetic's life as "short, disgusting, and painful."
Meanwhile, over in Rome, Galen (129 CE - c 200 CE), perhaps the most celebrated physician of western antiquity, indicated he had only seen a couple cases. This suggests that most diabetics from this era did not live long enough to be treated for their condition.
Ibn Sīnā, (980-1037 CE), known in the west as Avicenna, was a Persian physician, astronomer, scientist, philosopher and writer. He was born near Bukhara, in modern-day Uzbekistan, but traveled over much of the Persian world during a period of religious and political strife.

Aretaeus of Cappadocia
Avicenna produced over written 450 works in a variety of subjects, from optics to religion, math to poetry. Over half these works survive, including 40
on medicine.
He is considered the father of medicine in the Islamic Golden Age (roughly from the 8th to 14th centuries).
His book The Canon of Medicine, a fragment of which is shown to the right, became a standard medical text for over 600 years, and is still used in traditional middle-eastern medicine. In it, he
described the sweet taste of diabetic urine. He also described diabetic gangrene.

His treatment for diabetes used a mixture of lupine, turmeric, and fenugreek.
This mixture does, in fact, lower sugar levels, and is still a prescribed treatment in some
parts of the world.
Avicenna was also one of the first to differentiate between diabetes mellitus and
diabetes insipidus, the latter of which produces excessive urine, but has nothing to
do with high levels of sugar.
In his 58th year, Avicenna suffered from an intestinal issue and treated himself.
Someone suspiciously strengthened his prescription two and a half fold, which caused an intestinal ulcer. When he treated his discomfort with a mild opium, someone significantly strengthened this too.
Avicenna held on for a while, but eventually succumbed to the poison. He is buried in Hamadan, Iran.

Ibn Sinai (Avicenna)

Turmeric (Above)
and
Fenugreek (Left)


Cambridge-trained Nicholas Culpepper (1616-1654),
left, was considered a radical. He wrote pamplets
opposing the King, (Charles I), all priests and lawyers.
He also opposed high-priced physicians and what
he felt were harmful treatments like bloodletting
and expensive medicines. He despised the notion
that physicians should be able to control medical
knowledge and to charge high fees for dispensing
that knowledge. So he translated medical texts
from Latin to common English, and produced self-
help guides for the poor.
The College of Physicians opposed the publication
of these works, but they were sold very cheaply,
became very widely available, and have been in
publication ever since.
Culpepper married well and provided his services for free, and spent his time roaming the English countryside cataloguing medicinal herbs. Average people could get the information he advocated for free--a combination of herbal medicine and astrology.
Culpepper died of tuberculosis at 38. In his book "A Treatise on Aurum Potabile" (1657), published posthumously by his wife, Culpepper described making a tea of Goat's Rue (Galega Officianalis), above right, to treat diabetes. Nearly 300 years later, a chemical cousin of this plant was used to create the biguanide family of anti-diabetic medications, a family that includes Metformin.
.jpg)
London-based physician Thomas Willis (1621-1675), right, was a contemporary
of Culpepper, though Willis was trained at Oxford. Based on the knowledge
that the urine of diabetics was sweet, Willis added the word "mellitus" (Latin
for 'sweetened with honey') to the condition in his chapter in the anatomy
text Pharmaceutice Rationalis (1674), shown below. However, Willis believed
the sweetness was a result of acids, not sugar.
The same word, honey, had been used to describe
diabetes in ancient Indian medicine 2500 years earlier.
The word "mellitus" differentiated the condition
from diabetes insipidus, which is involves an increase
in urine, but is not associated with high sugar.
Willis was so well-known for his work with diabetes
mellitus that the condition was once also known as
Willis' disease. However, he was best known for his
text of 1664, Cerebri Anatome, which became the
definitive authority on neuroanatomy for the next
two centuries.
So important was that bookThomas Willis is considered today the Father of Neuroscience.
It was Willis who numbered the cranial nerves. This is the Willis after whom the 'Circle of Willis,'the arteries at the base of the brain, is named. (Every physician today is familiar with the Circle of Willis.)
Interestingly, Willis was part of an impressive network of scientists, physicians and scholars who frequently met to discuss cases that they handled and problems they had. This group included physicists Isaac Newton and Robert Hooke, the first modern chemist, Robert Boyle, physician and philosopher John Locke, physician and anatomist Richard Lower, and the architect and artist Christopher Wren. (This is a collection of intellect and talent that may never have been equalled, and if you're unfamiliar with any, they're each worth googling.)
At 54, Willis died of pleurisy, an inflammation of the membranes surrounding the lungs. The most common cause of pleurisy is a viral infection, though pancreatitis from diabetes can be another cause.
Willis is buried at Westminster Abbey.

Matthew Dobson (1735-1784) was born in Yorkshire, educated at the University of Edinburgh, right, and conducted a series of experiments on diabetes while working in Liverpool. In 1776, Dobson evaporated the urine of a diabetic and found (by tasting it) that sugar remained, thus discovering that diabetics had sugar in the urine.
Dobson also discovered that blood serum contained sugar, thus discovering hyperglycemia.
From this he deduced that the problem of sugar in the urine was not caused by the kidneys, but the sugar “previously existed in the serum of the blood.” And he realized that diabetes should be considered “a species of imperfect digestion and assimilation, the obvious indication of cure are, to strengthen the digestive powers” to assist with the assimilation of nutrients.
Dobson retired to Bath in 1780 and died of an unknown affliction four years later at age 49.
In 1788, an English physician named Thomas Cawley was performing an autopsy on a 34-year-old diabetic who he described as "strong, healthy and corpulent, accustomed to free living and strong corporeal exertions in the pursuit of country amusements." He developed diabetes, became emaciated and debilitated before succumbing to the disease. Dr. Cawley noted calcific stones and damage in the pancreas and a pancreas full of calculi, thus tying the disease to that organ.
In 1797, a Scottish military physician named John Rollo, right, became known for developing an effective diet for diabetes, when, in 1797, he noted the improvement in the health of an officer with diabetes when placed on a diet of meat and fat, low in grains and breads. This was the first low-carb diet designed for diabetics.

By the 19th century, doctors knew sugar worsened diabetes, and treatment was generally geared towards limiting dietary intake.
By that time, cadaveric dissections were not uncommon, and in 1869, a German medical student from Berlin named Paul Langerhans (1847-1888) found clumps of cells in the pancreas known today as Islets of Langerhans (below).
The Islets of Langerhans make up just 1-2% of the pancreas (a total of approximately 3 million of them in a healthy pancreas.) But they're vital for life, because some of the cells inside, cells known as beta cells, are what produce insulin.
Left image: Public Domain,
https://commons.wikimedia.org/w/index.php?curid=34177045
Right image: By Afferent - Own work, CC BY-SA 3.0,
https://commons.wikimedia.org/w/index.php?curid=28070032
The discovery of the Islets of Langerhans were not Langerhans' only contribution to medicine. When he was just an undergraduate student, he discovered a nerve cell in the skin. It took science over 100 years to figure out that these 'epidermal Langerhans cells' functioned as part of the immune system.


Langerhans was also instrumental in understanding the function of human macrophages. (To read a bit about macrophages' importance in our immune system and wound healing, visit our pages on infection and the effect of sugar on wound healing.) He was also involved in zoological research, authoring studies on a variety of amphibians.
Langerhans contracted tuberculosis in 1874, very likely from his work dissecting infected specimens at the University of Freiberg. The tuberculosis ultimately forced him to resign his position and relocate to the Portuguese island of Madeira, off the coast of Africa. In Madeira, he practiced medicine, studied his own disease, tuberculosis, and investigated the invertebrate animals of the region.
Langerhans died of kidney failure, a common complication of diabetes, five days short of his 41st birthday.


In 1889, the year after Langerhans' death, a couple of German
physicians based at the University of Strasbourg, then within the
borders of Germany, took another step forward in understanding
the condition of diabetes.
Joseph von Mering (1849-1908), shown on the left, and
Oskar Minkowski (1858-1931), to the right, removed the
pancreas from a dog. They found that the dog's urine began testing positive for sugar.
This experiment showed for the first time that the pancreas
was involved in the body's regulation of sugar.
Minkowski's brother Hermann was a renowned mathematician,
a specialist in geometry, and one of Einstein's teachers. His
other brother, Rudolph, was a renowned astronomer best known
for his work with understanding supernovae.
Mering later became known for his work on the therapeutic effects of barbiturates.
Sir Edward Albert Sharpey-Shafer (1850-1935), a British physician of a German immigrant father, was responsible for naming the substance responsible for diabetes mellitus as "insulin." The word was originally coined in 1914 as "insuline." 'Insula' came from the Latin word for islet (as in Islet of Langerhans, discussed above). The ending '-ine' is Latin for "of or pertaining to."
The word was later shortened to insulin, in 1922.
Sharpey-Shafer, considered the founder of endocrinology. Among his many professional achievements, he also coined the term 'endocrine,' and discovered the existence of adrenaline.

Image courtesy of Lippiatt - [1], CC BY 4.0, https://commons.wikimedia.org/w/index.php?curid=33147639
Eugene Lindsay Opie (1873-1971) was an American physician, a graduate of the first medical class at Johns Hopkins University.
In 1901, while at Johns Hopkins, Opie discovered which specific part of the pancreas was involved in diabetes, when he discovered that diabetes was caused by the destruction of the Islets of Langerhans In 1901.
After working at the Rockefeller Institute and Columbia in New York, then Washington University in St. Louis, Opie served in France in World War I. There he became interested in infectious disease, which killed more soldiers than did war wounds. He later worked at Penn, then Cornell, where he spent much of his time investigating tuberculosis--a major scourge of the early 20th century, and the disease that killed Culpepper and Langerhans.

Discovery of Insulin
So over the 45 years between 1869 and 1914, scientists learned that the Pancreas was important in the development of diabetes, that the specific cells involved with the metabolism of sugar were the newly-discovered Islets of Langerhans, and they coined a name, insulin, for the sugar-controlling substance produced by those cells.
The rush was now on to isolate insulin to see if it could be used to treat diabetes.
There was more than one group working on the project. But groups in Europe were hamstrung by the chaos, devastation, and overall lack of resources following the First World War.
This was not the case in Canada, however. And in 1921, a young Canadian surgeon at the University of Toronto, Frederick Banting, believed he had a surgical technique that would allow him to isolate the hormone insulin in dogs.

Frederick Banting (1891-1941)
Image in Public Domain
Banting approached John J.R. Macleod, a Scottish professor of physiology, then
at the University of Toronto, who had written a dissertation on diabetes.
Macleod was reluctant to the idea of working with Banting, as Banting had no
research experience. Nonetheless, Macleod was intrigued by Banting's proposal, and he agreed to oversee the work.
Banting offered the experimental facilities of his lab, along with the assistance of one of his students, Charles H. Best.

Banting and Best quickly set to work, and they were soon able to isolate and extract insulin through the technique envisioned by Banting.
Over the course of 1921, Banting and Best experimented with techniques to harvest enough insulin to perform their animal trials.
Their results were quite promising, but Macleod identified flaws with the design of the study, and demanded the study be repeated, with more experiments, more dogs, and superior equipment.
When the second round of experiments proved successful, Macleod helped get the work published.
While Banting were able to speed up the process of isolating insulin by switching his experiments from dogs to calves, then cows, neither Banting nor Best were biochemists by trade, and they still had difficulty producing and purifying enough product.
So Macleod called upon James Collip, once a student at the University of Toronto, but at that point Professor and Head of the Department of Biochemistry at the University of Alberta, to join their group in Toronto. Collip had been following the work of Banting, Best and Macleod, and he knew of their struggles in isolating pure insulin. He commented to a colleague that he could get the job done in two weeks.
Collip joined the team in December of 1921.
J. R. R. Macleod (1876-1935)
Image in Public Domain
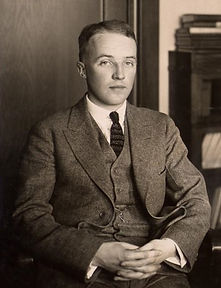
Charles H. Best (1899-1978)
Image in Public Domain
Days after Collip's arrival in Toronto, Banting, Best, and Macleod traveled to Yale University in Connecticut to present their findings at the conference for the American Physiological Society on December 30, 1921.

James Collip (1892-1965).
Image in Public Domain

Above: Yale University. Image in Public Domain.
Below: An abstract submitted for their work.
As lead researcher, Banting was to make the presentation. But Banting was nervous, and was not a natural speaker. In fact, Banting gave such a poor delivery of his paper that the presentation was a near disaster, and the audience in attendance became quite vociferous in their criticism of Banting's work.
Macleod, who was the chair of the session, stepped in to the discussion to support his colleague. He spoke at length in response to the withering criticism being leveled at Banting and his work.
Instead of being appreciative of Macleod's intercession on his behalf, Banting was furious. He viewed Macleod as trying to sideline him and take credit in the scientific community for the discovery of Insulin.
The experience made Banting immediately suspicious of Collip, too. To Banting, Collip appeared to be someone hired by Macleod to take the production of insulin away from him.
Alarmed, Banting and Best quickly set to work in a race to beat Collip and Macleod--who they now saw as competitors--in a race to isolate insulin.
Unfortunately for Banting, it wasn't much of a contest. In less than a month of his arrival in Toronto, Collip, whose very specialty was this sort of task, found a method to produce pure insulin, using a 90% alcohol solution.
Despite the enormous success of quickly achieving precisely the results he was asked to produce, Collip suddenly found himself in the middle of a very prickly situation with Banting and Best, who were suspicious and resentful of his very presence. Uncomfortable in dealing with Banting and Best, Collip informed them that he was more comfortable sharing his results with Macleod.
This enraged Banting. Why wouldn't he share his results with the actual discoverers of insulin? To Banting, this was clear proof Collip was hired by Macleod to steal his discovery. So furious was Banting, that it is said that Best actually had to restrain him from attacking Collip.
Passions were high, but there was too much at stake. A truce was quickly arranged, and human experiments were begun with the purified insulin.
The results of the human trials of insulin were remarkable. Comatose patients were revived. Those near death rallied.
One of the first treated patients was the 8-year-old aunt of the author of this web page, who was quite ill and also experienced a huge improvement in her health with this new drug, insulin.

By all accounts, insulin was a huge success. And on May 3, 1922, the group again made a presentation of their findings to an international medical conference of the Association of American Physicians in Washington, DC--this time with Macleod as the presenter. At this point, there was a very positive reaction from the audience.
The work was seen as so important that the 1923 Nobel Prize in Physiology or Medicine was to awarded to the discoverers of insulin. But who would receive the award? Banting, who came up with the idea of how to isolate insulin? Best, who assisted with the entire project? Macleod, who oversaw the project, provided the space and materials, demanded clearer research? What about Collip, who was needed to develop a pure product so trials could be run on people?
1923 The Nobel Prize in Medicine or Physiology
In the end, the Nobel Committee bestowed the award to
Banting and Macleod. Best and Collip were left out.
While Banting would be expected to have been thrilled to
become the youngest person to ever win a Nobel Prize in
Medicine or Physiology, (he was 31 at the time), Banting
was livid.
To Banting, the Nobel Committee's inclusion of Macleod
and exclusion of Best's contribution was further proof of
Macleod's duplicity.
Knowing the importance of Best's work, Banting shared
his Nobel Prize money with Best.
In turn, recognizing Collip's vital contribution in the
production and purification of insulin, Macleod shared his
Nobel Prize money with Collip.

Patent for Insulin
Banting, Best and Collip shared the patent for insulin. The patent could have made them rich.
But they felt it would be unseemly to profit at the
expense of their patients' infirmity. So they sold the patent to the University of Toronto for one dollar each.

Aftermath
After the Nobel Prize was awarded, Banting and Macleod never again spoke.
The reserved and modest Macleod was anxious to remove himself from the drama, and surely achieved his goal when he found a position in New Brunswick to work with insulin in fish.
By 1929, Macleod moved even further away from Toronto, when he returned to Scotland, becoming Dean of the University of Aberdeen Medical Faculty. He continued work as a researcher, lecturer and an author of over 200 research papers and eleven text books.
Macleod developed debilitating rheumatoid arthritis, however, and died of the condition in 1935 at age 58.
Banting and Best stayed at the University of Toronto. Banting, who remains to this day the youngest ever recipient of the Nobel Prize in Medicine or Physiology, changed the focus of his research to a variety of disparate topics, from cancer, to the effects of mustard gas, to aviation medicine.
He died in a plane crash in Newfoundland in 1941. He was 49.
James Collip soon returned to his university position at the University of Alberta in Edmonton. He later moved on to McGill University in Montreal to become the Chair of Biochemistry.
Collip ended his career as the Dean of the University of Western Ontario in London, Ontario.
Collip was a pioneer in endocrinology, and he is particularly known for his work with parathyroid hormone.
He died of a stroke in 1965 at age 72.
Charles Best succeeded Macleod as the Chair of Physiology at the University of Toronto.
Of the four-member group, Best passed away last, in 1978 at age 79. Through the years, Best promoted the story of how he and Banting developed insulin, diminishing the contributions of Macleod and Collip. As a result, Macleod's public imaged remained tarnished for years.
Wishing to avoid delving into the controversy, the University of Toronto kept much of the documentation on the story hidden until Best's death.
As documentation became more public, it's become more accepted that Banting and Best's story was biased and distorted, and each of the four figures had important roles in the development of insulin.
Oral Medications
The discovery Insulin was an enormous breakthrough in the treatment of diabetes. However, the push was soon on to ditch the needles and find an oral alternative.
The first two classes of oral medications for diabetes were the biguanides and the sulfonylureas.
Biguanides are related to the chemical guanidine found in the plant Goat's Rue, Galega Officinalis, which, as discussed above, Nicholas Culpepper argued for three centuries earlier.
Galega officinalis was first described in the scientific literature in 1922, and its
sugar-lowering effects were noticed in 1929. But the drug was soon forgotten
until it was rediscovered by Jean Sterne of Paris in the 1950s. Sterne, seen to
the right, was the first to try the medication metformin on humans and he
coined the trade name Glucophage (sugar eater) for the medication. It became
available in the UK in 1958 and in Canada in 1972 and the US in 1994.
Today the drug is the first-line medication for most newly-diagnosed diabetics.
It's generally considered safe, it lowers blood sugar effectively, it protects your
heart, it lowers your weight, it doesn't interact with many mediations, and it's
quite inexpensive.

While experimenting to see whether a drug called sulfonylurea might be an
effective treatment for typhoid in 1942, Dr. Marcel Janbon (near right) of
Montpellier, France found that the dogs became ill and sometimes died.
Investigation found that the drug caused the dogs' blood sugar to plummet.
Enlisting the physician August Loubatieres (far right), they experimented with diabetic patients, and found the same effect. Subsequent experiments proved that sulfonylurea caused the pancreas cells to release insulin.
Sulfonylureas are effective, but they can lower blood sugar excessively, and they can cause weight gain.
Today there are many diabetic medications in this class. Interestingly, several also act as herbicides because they interact with a plants ability to synthesize amino acids.


Today we know diabetes mellitus as a group of metabolic conditions of high sugar levels in the blood. It is still known to bring frequent urination, increased thirst and hunger, and muscle wasting.
We categorize most diabetes as one of these three:
Type 1 Diabetes mellitus is the condition where the pancreas doesn't produce enough insulin.
Type 2 Diabetes is a result of "insulin resistance," where the cells in the pancreas begin to fail to respond to insulin. This is typically a result of obesity.
(Type 1 Diabetes has sometimes been known as "Insulin-dependent Diabetes" or "Juvenile Diabetes," and Type 2 has sometimes been known as "Non-Insulin-Dependent Diabetes or IDDM" or "Adult-Onset Diabetes." However, these names are inaccurate. It is quite possible for an adult to be a Type 1 diabetic or a Type 2 to require insulin, so describing diabetes by age or insulin usage is inaccurate.)
A third well-known form of diabetes, Gestational Diabetes, develops during pregnancy, but tends to resolve after childbirth.
There are, in fact, several other forms of diabetes. For example, there's drug-induced diabetes (such as that caused by steroid usage), infection-induced diabetes (for example, that caused by rubella or German measles), and diabetes as a result of pancreatic cancer or cystic fibrosis). But the result of each form of Diabetes Mellitus is difficult-to-control sugar levels, which can have a significant impact on the body.
To return to the top of the page,
click on the leaf to the right.
This page written by Dr. S A Schumacher
Podiatric Surgeon
Surrey, British Columbia Canada
Unless otherwise indicated, all clinical
photographs owned and provided by
Dr. S A Schumacher.
They may be reproduced for educational
purposes with attribution to:
Dr. S A Schumacher
.webp)
